It is that time of year again when The Centers for Medicare and Medicaid Services (CMS) publishes their for the upcoming year. Comments this year are due no later than June 7th. The Proposed Rule will become final later this year with an effective date of October 1, 2021. Here is a very high overview of what is in this Fiscal Year (FY) 2022 Propose Rule.
| TOPIC | FY 2022 PROPOSAL |
| SNF Proposed Payment Updates | SNF PPS Payment Update of 1.3. This is a $444 million increase compared to FY2021.2.3% market basket increase factor, minus the 0.8% forecast error correction, an additional 0.2%-point reduction for the multifactor productivity adjustment, and $1.2 million decrease for the proposed reduction to SNF PPS rates for the recent blood-clotting factors exclusion discussed in more detail below.Does Not Include the FY 2022 SNF VBP Reductions estimated to be $184.25 million. |
| Updated Base Rates for PDPM Components | *Unadjusted federal rate per diem for urban and rural prior to adjustment for case-mix Table 4: FY 2022 Unadjusted Federal Rate Per Diem – URBAN Rate Component PT OT SLP Nursing NTA Non-Case-Mix Per Diem Amount $62.84 $58.49 $23.46 $109.55 $82.64 $98.10 Table 5: FY 2022 Unadjusted Federal Rate Per Diem – RURAL Rate Component PT OT SLP Nursing NTA Non-Case-Mix Per Diem Amount $71.63 $65.69 $29.56 $104.66 $78.96 $99.91 |
| Changes in SNF PPS Wage Index | Continue using the hospital inpatient wage data to develop a wage index to be applied to SNFs. The FY2022 wage index is found in Tables A and B available on the CMS website here: CMS is adopting the latest OMB Bulletin geographic delineations; no specific wage index updates noted as a result |
| Patient-Driven Payment Model (PDPM) | Parity Adjustment Recalibration: due to PDPM data being different from what the projected (PDPM not budget neutral). CMS states that the FY 2020 parity adjustment may have inadvertently triggered a significant increase in overall SNF payments. CMS feels that there have been significant changes observed in the SNF utilization under PDPM and changes are not due to the pandemic. CMS is seeking comments on how to move forward for recalibrating the PDPM parity adjustment to ensure budget neutrality. If adjustment is implemented, CMS would consider multi-year reduction implementation. CMS’ analysis resulted in PDPM parity adjustment factor decrease from 46% to 37% for each PDPM case-mix adjusted components. If this were to be applied in FY 2022, CMS estimates this would result in a 5.0% SNF reduction in spending ($1.7 billion). PROPOSED methodology and adjustment recalibrated CMI rates 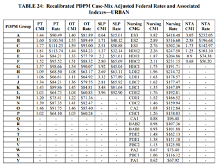 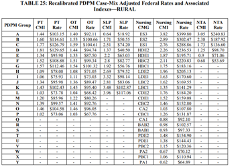 |
| PDPM ICD-10 Coding Update | Two New ICD-10 Codes Related to Sickle-Cell Disease: Change assignment from “Medical Management” to “Return to Provider”Three New ICD-10 Codes Related to Esophageal Conditions: Change assignment of these codes from “Return to Provider” to “Medical Management”ICD-10 Code M35.81: Multisystem inflammatory syndrome- change assignment from “Non-Surgical Orthopedic / Musculoskeletal” to “Medical Management”;Three New ICD-10 Codes: Types of neonatal cerebral infarction- P91.821- Neonatal cerebral infarction, right side of brain, P91.822- Neonatal cerebral infarction, left side of brain, and P91.823- Neonatal cerebral infarction, bilateral, – change from “Return to Provider” to “Acute Neurologic” because while a neonate is unlikely to be a Medicare beneficiary, this diagnosis could continue to be used later in life.ICD-10 Code on Vaping-Related Disorder: U07.0- Vaping-related disorder- change from “Return to Provider” to “Pulmonary” in recognition that this condition could be the primary diagnosis for a SNF stay.Request Made to CMS Regarding ICD-10 Code G93.1: Anoxic brain damage, not elsewhere classified- change from “Return to Provider” to “Acute Neurologic”. CMS reviewed the recommendation and is proposing to make this change as requested. |
| SNF Quality Reporting Program (QRP) | Request for Information (RFI): Closing the Health Equity Gap.SNF Healthcare-Associated Infections (HAI) Requiring Hospitalization Measure: Beginning FY 2023 SNF QRP addition: claims-based measure.COVID-19 Vaccination Coverage Among HealthCare Personnel Measure Beginning FY 2023 SNF QRP: Would require SNF to report on COVID-19 vaccination via the CDC and Prevention National Healthcare Safety Network beginning 10/1/21.Transfer of Health (TOF) Information to the Patient: PAC Quality Measure- update the denominator definition to avoid counting the patient in both TOH measures (TOH Information to the Patient-PAC; TOH information to patients discharged home under home health or hospice).Public Reporting of QMs with Fewer than Standard Numbers of Quarters Due to COVID-19 PHE Exemptions: Proposing to update the number of quarters used for public reporting to account for the SNF QRP quarter exceptions during the PHE. |
| SNF Value-Based Purchasing (VBP) Program | Proposal to suppress the SNF readmission measure for FY 2022 VBP Program due to COVID-19 PHE. Expanded SNF VBP Program: Considering the addition of measures, regardless of payer: functional status, patient safety, care coordination, or patient experience. |
| New Blood Clotting Factor Exclusion from SNF Consolidated Billing | Proposing a proportional reduction in the Medicare Part a SNF rates to account for a new exclusion of specified blood clotting factors for the treatment of patients with hemophilia or other bleeding disorders from the consolidated billing requirement under the SNF PPS. Would impact items and services provided on or after October 1, 2021. If finalized, would result in a decrease of approximately $1.2 million in SNF spending, which offsets the increase in Part B spending that would occur from this change. |
| Health IT | Office of the National Coordinator for Health Information Technology (ONC) Cures Act Final Rule: Information blocking workgroup between CMS and ONC to facilitate collaboration to develop standards.Advancing Health Information Exchange Request for Information on the use of Fast Healthcare Interoperability Resources (FHIR). |
